on the April 7, 2026
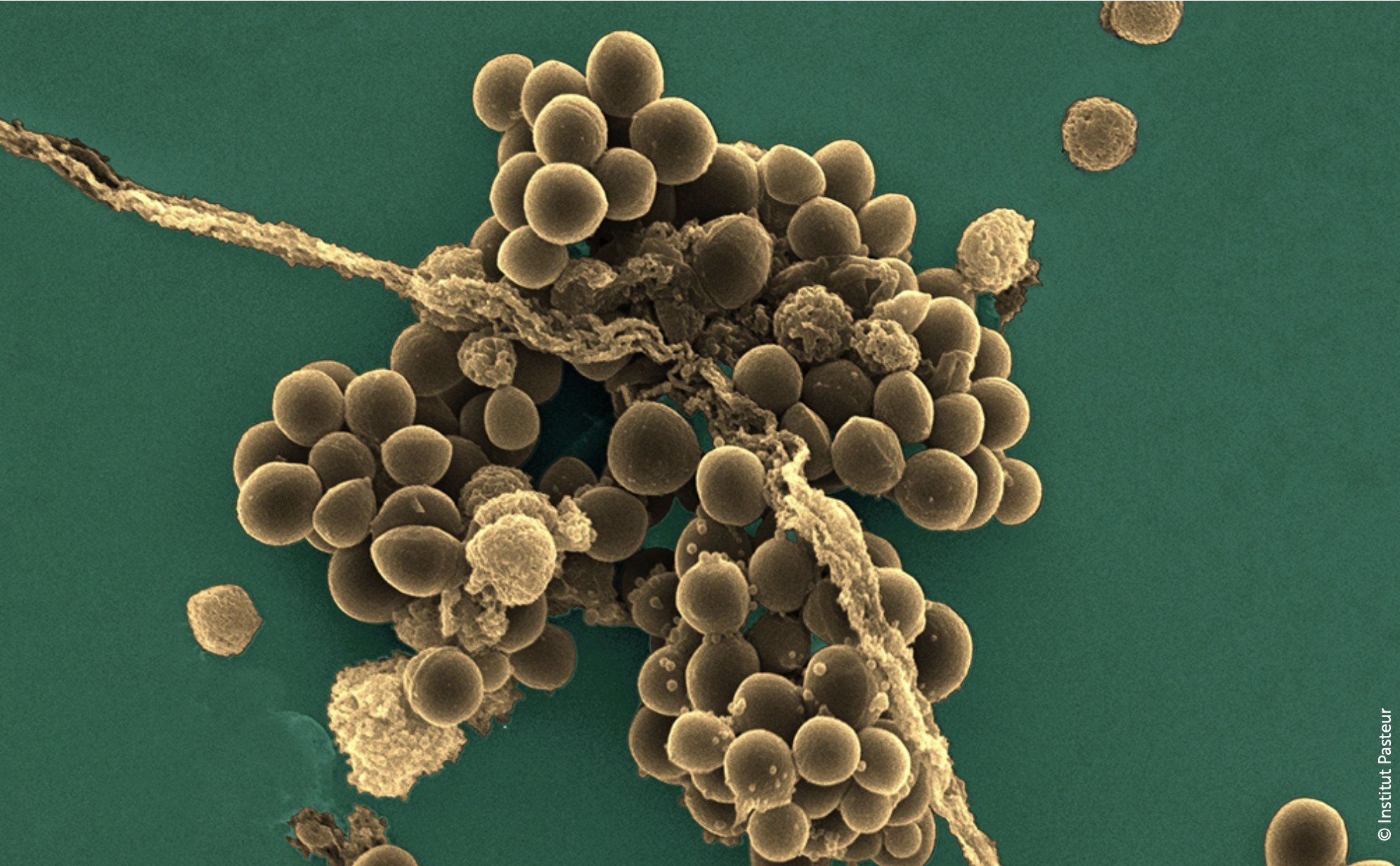
Every year, bacteria become more resistant to our medications. At the Institute of Chemistry in Nice, researcher Cyril Ronco is taking on a formidable foe: methicillin-resistant Staphylococcus aureus (MRSA). His team is developing new molecules to prevent the therapeutic deadlock that is already threatening hospitals.
Drug resistance is often portrayed as a problem of the future. In reality, it already exists. When we try to eliminate a living system—a bacterium or a cancer cell—it does everything it can to survive. The result: over time, these living systems mutate, and certain antibiotic or anticancer treatments become ineffective.
This is the context for Cyril Ronco’s work. His goal: to design new molecular tools capable of circumventing these resistance mechanisms. Among the most urgent threats, Staphylococcus aureus is the most common cause of nosocomial infections.
This formidable bacterium causes serious infections and is one of the leading causes of hospital-acquired infections.
The problem? Some strains are now multidrug-resistant, meaning they are resistant to nearly the entire arsenal of available antibiotics. In practice, doctors use a “first-line” treatment, then a second, then a third… For Staphylococcus aureus, some patients unfortunately reach the end of the list, facing treatment failure.
A NEW IDEA BORN FROM DIALOGUE BETWEEN CHEMISTRY, BIOLOGY, AND CLINICAL PRACTICE
The project led by Cyril Ronco emerged from close collaboration between his team of chemists and that of Laurent Boyer, a researcher at the Mediterranean Center for Molecular Medicine (C3M), in direct consultation with clinicians at the Hôpital de l’Archet.
Faced with this impasse, innovation is needed. The initial idea is ingenious: to create a new type of antibacterial agent by fusing two distinct chemical elements.
One is very common in existing antibiotics; the other is rarer and particularly versatile. By combining them, the researchers hope to obtain a completely novel chemical structure capable of circumventing known resistance mechanisms.
To test this hypothesis, more than 300 different molecules incorporating this fusion structure were first synthesized in the laboratory. They
were then tested against two strains of Staphylococcus aureus.
The verdict was harsh: only two molecules belonging to the same chemical family showed promising activity. In research, these are called “hit” molecules.
But this is only the beginning. Starting with these hit molecules, the chemists begin a meticulous optimization process. They modify each part of the molecule, one chemical group at a time, to understand what triggers the antibacterial activity and how to amplify it. This approach allows for establishing correlations between a 3D structure and the protein’s activity: a true mapping of molecular activity.
FROM THE LAB TO THE MOUSE... AND BEYOND
Through these successive adjustments, researchers obtain lead molecules—molecules that are more active and more selective against the therapeutic target. They are then subjected to a series of essential tests: absence of toxicity to human cells, efficacy against clinical strains of Staphylococcus aureus collected from patients, including multidrug-resistant strains.
The results are very encouraging. The compounds are active against both “naive” bacteria and bacteria already resistant to multiple antibiotics. Better yet: no resistance appears to develop during prolonged experiments. This is a rare achievement, which can be explained by the entirely novel nature of this chemical class.
In vivo testing is continuing in mice. Here again, the results are promising: the molecules are soluble, well distributed throughout the body, and effective against infection, with properties comparable to those of drugs already on the market.
Now, a key step remains: the transition to industrial development. The project led to the filing of a patent in 2022 protecting the family of molecules and their activity. As a winner of the i-Lab national innovation competition, the project has sufficient funding, and a startup is being established to carry out the rest of the development: from preclinical phases through to clinical trials.
Alongside Laurent Boyer and Juan Garcia-Sanchez, Cyril Ronco is now preparing for the next steps. These molecules need to be formulated so they can become actual treatments: cream or tablet form, large-scale production, stability testing... On the way to a future drug!



